Index of Species Information
SPECIES: Tilia americana
|
|
 |
| American basswood fruits. Creative Commons image by Chris Evans, University of Illinois, Bugwood.org. |
Introductory
SPECIES: Tilia americana
AUTHORSHIP AND CITATION:
Sullivan, Janet. 1994. Tilia americana. In: Fire Effects Information System, [Online].
U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station,
Fire Sciences Laboratory (Producer). Available:
https://www.fs.usda.gov/database/feis/plants/tree/tilame/all.html [].
Revisions:
Infrataxa and citation [70] were added on 17 July 2014. The common name was changed in FEIS
from: bassswood to: American basswood. Images were added on 3 March 2018.
ABBREVIATION:
TILAME
TILAMEA
TILAMEC
TILAMEH
SYNONYMS:
For Tilia americana L. var. americana:
T. americana var. neglecta (Spach.) Fosberg [47]
For Tilia americana L. var. caroliniana:
Tilia floridana Small
For Tilia americana L. var. heterophylla:
Tilia heterophylla Vent.
Tilia michauxii Nutt.
NRCS PLANT CODE:
TIAM
TIAMA
TIAMC
TIAMH
COMMON NAMES:
American basswood
basswood
linden
TAXONOMY:
The scientific name for American basswood is Tilia americana L. [47,29].
Varieties include [70]:
Tilia americana L. var. americana, American basswood
Tilia americana L. var. caroliniana (Mill.) Castigl., Carolina basswood
Tilia americana L. var. heterophylla (Vent.) Loudon, American basswood
LIFE FORM:
Tree
FEDERAL LEGAL STATUS:
No special status
OTHER STATUS:
NO-ENTRY
DISTRIBUTION AND OCCURRENCE
SPECIES: Tilia americana
GENERAL DISTRIBUTION:
The native range of American basswood extends from southwestern New Brunswick and
Maine west to southern Quebec, southern and western Ontario, Michigan,
Minnesota, and southeastern Manitoba; south to eastern North
Dakota, northern and eastern Nebraska, eastern Kansas, and northeastern
Oklahoma; east to northern Arkansas, Tennessee, western North Carolina,
and New Jersey [47].
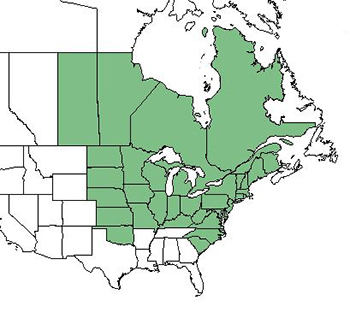 |
 |
 |
| Distributions of Tilia americana var. americana, Tilia americana var. caroliniana, and Tilia americana var. heterophylla, respectively. Area shaded in blue denotes introduced range. Maps courtesy of USDA, NRCS. 2018. The PLANTS Database.
National Plant Data Team, Greensboro, NC [70] [2018, March 3]. |
ECOSYSTEMS:
FRES10 White - red - jack pine
FRES15 Oak - hickory
FRES17 Elm - ash - cottonwood
FRES18 Maple - beech - birch
FRES19 Aspen - birch
STATES:
AR CT DE IL IN IA KS KY ME MD
MA MI MN MO NE NH NJ NY NC ND
OH OK PA RI SC SD TN VT VA WV
WI MB NB ON PQ
BLM PHYSIOGRAPHIC REGIONS:
NO-ENTRY
KUCHLER PLANT ASSOCIATIONS:
K081 Oak savanna
K095 Great Lakes pine forest
K099 Maple - basswood forest
K100 Oak - hickory forest
K101 Elm - ash forest
K102 Beech - maple forest
K103 Mixed mesophytic forest
K104 Appalachian oak forest
K106 Northern hardwoods
K107 Northern hardwoods - fir forest
K108 Northern hardwoods - spruce forest
SAF COVER TYPES:
1 Jack pine
16 Aspen
20 White pine - northern red oak - red maple
21 Eastern white pine
23 Eastern hemlock
24 Hemlock - yellow birch
25 Sugar maple - beech - yellow birch
26 Sugar maple - basswood
27 Sugar maple
28 Black cherry - maple
39 Black ash - American elm - red maple
42 Bur oak
52 White oak - black oak - northern red oak
55 Northern red oak
58 Yellow-poplar - eastern hemlock
59 Yellow-poplar - white oak - northern red oak
SRM (RANGELAND) COVER TYPES:
NO-ENTRY
HABITAT TYPES AND PLANT COMMUNITIES:
American basswood generally occurs in mixed stands and rarely forms pure stands
[16]. It is codominant in the sugar maple (Acer saccharum)-American basswood
cover type, and is a common component of many other mesophytic forests [24].
Associates in the sugar maple-American basswood type include white ash (Fraxinus
americana), northern red oak (Quercus rubra), eastern hophornbeam
(Ostrya americana), red maple (A. rubrum), and American elm (Ulmus
americana) [16,24]. Typical sugar maple-American basswood communities in
Wisconsin and Minnesota include 21 percent northern red oak, 35 percent
American basswood, 17 percent sugar maple, and 17 percent other species [68]. To
the east, eastern hemlock (Tsuga canadensis) may be present, and
communities on mesic sites would be more like the following: 52 percent
sugar maple, 27 percent American basswood, 14 percent yellow birch (Betula
alleghaniensis), 4 percent eastern hemlock, and 3 percent other species [68].
Publications in which American basswood is listed as a dominant, codominant,
or indicator species include:
The vegetation of Wisconsin [17]
The principal plant associations of the Saint Lawrence Valley [18]
Deciduous forest [31]
A forest classification for the Maritime Provinces [49]
A classification of the deciduous forest of eastern North America [53].
MANAGEMENT CONSIDERATIONS
SPECIES: Tilia americana
WOOD PRODUCTS VALUE:
American basswood wood is soft and light; it is valued for hand carving and has
many other uses including cooperage, boxes, veneer, excelsior, and pulp
[15,16]. American basswood is economically important for timber, especially in
the Great Lakes States [16].
Carey and Gill [11] rated American basswood as fair (their lowest rating) for
firewood.
IMPORTANCE TO LIVESTOCK AND WILDLIFE:
American basswood is preferred browse for white-tailed deer [67]. American basswood
flowers are visited by honeybees for nectar [16]. The easily decayed
wood produces a disproportionate number of cavities which are used by
cavity-nesting animals including wood ducks [21], pileated woodpeckers
[34], other birds, and small mammals [11]. Carey and Gill [11] rate
American basswood as fair (their lowest rating) for all wildlife.
PALATABILITY:
NO-ENTRY
NUTRITIONAL VALUE:
NO-ENTRY
COVER VALUE:
NO-ENTRY
VALUE FOR REHABILITATION OF DISTURBED SITES:
American basswood is of limited use in mixed hardwood plantings on disturbed
sites in Ohio [75].
OTHER USES AND VALUES:
American basswood is planted as a shade tree or ornamental [16]. The fibrous
inner bark ("bast") has been used as a source of fiber for rope, mats,
fish nets, and woven baskets [15,16].
OTHER MANAGEMENT CONSIDERATIONS:
American basswood stump sprouts can be managed for sawtimber [16]. The number of
stump sprouts declines with the age and size of the cut trees [7].
Since sprouts originating at or below the ground line are more resistant
to butt rot, stumps should be cut very close to the ground [46] or
burned [57]. Early thinning of sprouts is needed to ensure good quality
and rapid growth [16]. In West Virginia an average of 14.4 7-year-old
stump sprouts per clump was removed to allow good growth of the
remaining one or two stems [45]. Larger stems (greater than 51.7 inches
[1.31 m] d.b.h. will also respond to release [63].
Direct seeding may also have potential for good American basswood regeneration
[20]; natural seed reproduction can be encouraged by shelterwood cutting
systems [16]. Shelterwood systems also enhance American basswood production
where advance regeneration is large enough to compete successfully with
other hardwoods and shrubs [7]. Advance growth should be 2 feet (0.6 m)
or more in height before the overstory is harvested [42]. Sites that
are harvested to 60 percent residual stems may need to be cut again to
allow the best growth of northern hardwoods (including American basswood) [55].
Even-aged management systems can also be used successfully for American basswood,
particularly where sprouts have been properly thinned [23].
American basswood and other tolerant species are often established in the
understory of oak (Quercus spp.)-hickory (Carya spp.) stands, and tend
to take over a site when the overstory is harvested, regardless of the
silvicultural system used [61]. American basswood invasion of mixed oak stands
is a problem for oak regeneration in Wisconsin [13].
High densities of white-tailed deer can result in American basswood seedling
height growth reduction or even complete loss of American basswood from the stand
due to overbrowsing [67]. On old-field sites American basswood is often subject
to damage from mice and voles girdling the stems. Rabbits also feed
heavily on seedlings and small saplings. Seed predators include mice,
squirrels, and chipmunks [16].
Insect pests of American basswood include the linden borer (which damages weak,
very young, or "overmature" trees) and the following defoliators: linden
looper, American basswood leafminer, spring cankerworm, fall cankerworm,
white-masked tussock moth, gypsy moth, and forest tent caterpillar.
None of these pests is considered a serious threat [16]. American basswood is
easily decayed by fungi, and butt rot is an important factor in loss of
merchantable timber [16].
American basswood is susceptible to many herbicides, but is resistant to 2,4-D
and 2,4,5-T [8].
BOTANICAL AND ECOLOGICAL CHARACTERISTICS
SPECIES: Tilia americana
GENERAL BOTANICAL CHARACTERISTICS:
American basswood is a native deciduous tree. Mature heights range from 75 to
130 feet (23-40 m) with diameter ranges from 36 to 48 inches (91-122 cm)
[16]. The bark of mature trees is up to 1 inch (2.54 cm) thick at the
base of the trunk [2,15]. The bark is furrowed into narrow,
flat-topped, firm ridges with characteristic horizontal cracks; young
trees have smooth, thin bark [15]. The inflorescence is a drooping
axillary cyme. The fruit is dry, hard, indehiscent, subglobose to
short-oblong, and is usually 0.2 to 0.28 inch (5-7 mm) in diameter, and
bears one or two seeds [29].
The root system of American basswood is composed largely of lateral roots; it
does not usually form a taproot [16]. American basswood root depths are usually
shallow relative to associated species root depths. In prairie soils
bur oak (Quercus macrocarpa), shellbark hickory (Carya lacinosa), and
northern red oak all have deeper roots than does American basswood. Maximum root
depth in American basswood averages 1.2 feet (36.5 cm) the first year, and 5 to 6
feet (1.5-1.8 m) by the third year; roots are not well developed below
approximately 2 feet (61 cm) [73]. On prairie soils the deepest roots
of a 28-year-old American basswood were 27 feet (8.2 m) but most of the roots
were in the top 4 to 5 feet (1.2-1.5 m) of soil [74]. Adventitious
roots will develop as the stem is buried, as occurs on the sand dunes
near southern Lake Michigan [16].
The tree crown is usually broad and rounded, but in close stands is more
columnar. The branches are small, weak, and often pendulous [2].
Maximum longevity is approximately 200 years [16].
RAUNKIAER LIFE FORM:
Phanerophyte
REGENERATION PROCESSES:
The youngest recorded age at which American basswood first reproduces is 15 years
[48]. American basswood flowers are insect pollinated, mostly by bees and flies
[16]. American basswood produces good quantities of seed at 1- to 3-year
intervals [16,30]. The relatively heavy fruits are not usually carried
long distances by the wind [42,52]. Seeds can remain dormant for up to
3 years [42]. This deep dormancy is thought to be caused by an
impermeable seedcoat, dormant embryo, and tough pericarp. Acid
scarification and cold stratification enhance seed germination on
mineral soil [16,42,58], but few seeds actually germinate under normal
conditions [16].
Shade enhances establishment and initial survival, but heavy shade
limits subsequent growth and development [16]. Seedlings can establish
in as little as 25 percent of full sunlight [55]. The higher soil
temperatures in forest openings are better suited for good seedling
growth [16], but seedlings are sensitive to soil nutrient deficiencies
which may render them less tolerant to shade than older trees [69].
However, dense reproduction is only obtained under partial canopies [42].
Most American basswood reproduction originates as stump sprouts. Almost all
American basswood trees 4 inches (10 cm) or less d.b.h. will sprout from the
stump, and more than 50 percent of sawlog-size trees will sprout as well
[16]. Sprouts have been obtained from American basswood trees over 100 years
old; 57 percent of trees 20 inches [50.8 cm] or more in diameter
sprouted [46].
SITE CHARACTERISTICS:
American basswood is characteristically found in rich uplands on mid-slopes in
mixed deciduous forests. It is sometimes found in swamps [71].
American basswood is generally confined to sandy loams, loams, or silt loams, and
achieves its best growth on the finer textured soils. Best growth is on
mesic sites, but American basswood will also grow on coarse soils that are well
drained. In Wisconsin American basswood exhibits a bimodal distribution with
respect to moisture; it achieves peak importance values on wet-mesic
sites and on dry-mesic sites, but is "outcompeted" by sugar maple on mesic
sites [17]. In southern Wisconsin, maple-American basswood forests are largely
restricted to northern exposures [2]. American basswood is moderately tolerant
of flooding; it occurs on floodplain sites that have probabilities of
annual flooding between 50 and 100 percent [54]. Acceptable soil pH
ranges from 4.5 to 7.5, though American basswood occurs most often on less acidic
to slightly basic soils. Because American basswood is nitrogen demanding it
grows poorly on nitrogen deficient soils [16].
At the western limits of its range, American basswood usually grows on the
eastern side of lakes and along major drainages where it is naturally
protected from fire. American basswood is commonly found in ravines and
protected, moist sites at the prairie-woodlands interface in Nebraska
[2]. In North Dakota and Minnesota American basswood achieves its highest
densities on intermediate slopes; it is found in lower numbers on both
the drier uplands and the wet bottomlands [72]. According to Crow [16],
American basswood's distribution is more closely associated with edaphic and
moisture conditions than with fire.
The maximum elevation at which American basswood is found is 4,930 feet (1500 m)
in the southern Appalachian Mountains [22].
SUCCESSIONAL STATUS:
Facultative Seral Species
American basswood is moderately tolerant of shade. It achieves its highest
densities in sugar maple-American basswood stands that are late successional to
climax forests. Sugar maple-American basswood can eventually replace oak-hickory
on favorable upland sites in the upper Midwest. Succession of
oak-hickory to sugar maple-American basswood can be accelerated where harvesting
or other disturbance releases the tolerant understory species [40].
Braun [9] classified American basswood as a member of the regional climax forest
in Ohio that also contains American beech (Fagus grandifolia), ash
(Fraxinus spp.), sugar maple, and yellow-poplar (Liriodendron
tulipifera). In eastern Nebraska northern red oak and American basswood are
described as climax dominants at the western limits of the forest [2].
In New York American basswood is a subclimax species, where it may increase in
importance after heavy logging [6]. In Minnesota sugar maple-American basswood
communities are climax on fine mineral soils. In Itasca State Park,
Minnesota, succession from prairie to white pine (Pinus strobus)-bur oak
to sugar maple-American basswood is explained by fire exclusion and/or increased
moisture. The authors concluded available evidence supports the climate
model better than the fire exclusion model [10].
The persistence of the moderately shade tolerant American basswood in stands
containing highly shade tolerant sugar maple is dependent on their
differing modes of reproduction. Sugar maple produces large numbers of
seedlings which are positively correlated with the occurrence of
American basswood in the canopy [27]. Stump sprouting allows American basswood to
maintain itself in a stand with the more shade-tolerant sugar maple;
American basswood stump sprouts can reach canopy size faster than the more
numerous maple seedlings [16,27,71].
Patchy or large scale disturbance may favor American basswood because of its
sprouting ability and presence in the understory. American basswood increased in
relative importance value after a tornado caused severe damage to a
sugar maple-Ohio buckeye (Aesculus glabra) stand in Indiana. The source
of the increase was not specified by the authors; it may have come from
stump sprouts and/or released individuals, coupled with the loss of
other species [51].
SEASONAL DEVELOPMENT:
American basswood usually flowers in June, but flowering dates range from late
May to early July [16]. Flowering occurs from 1 to 4 weeks after spring
leaf-out [1,16]. In Minnesota, bud swell occurs in from late April to
early May, and leafing out occurs from early to mid-May. Seeds are
dispersed in October, and leaf fall occurs from September to October [1].
FIRE ECOLOGY
SPECIES: Tilia americana
FIRE ECOLOGY OR ADAPTATIONS:
American basswood is rated as a fire sensitive species; the thin bark is easily
damaged by fire, as are the shallow roots [16]. However, American basswood that
has been top-killed by fire will sprout vigorously from the root crown [71].
American basswood is most common in forests with long fire-free intervals. Fire
suppression has apparently encouraged its increase. In the absence of
fire, paper birch (Betula papyrifera)-eastern white pine-bur oak forests
are being replaced in by American basswood, balsam fir (Abies balsamea), and
black ash (Fraxinus nigra) in the north-central states [1]. American basswood
and sugar maple usually replace postfire aspen stands (Populus
tremuloides and P. grandidentata) on rich, moist sites in Minnesota
[33]. The presence of sugar maple-American basswood stands is correlated with
longer fire-free intervals in Minnesota; pines (including white pine,
red pine [Pinus resinosa] and jack pine [P. banksiana]) and aspens
occur on sites that burn more frequently [14]. In jack pine or red
pine-white pine stands which experience frequent fire, American basswood is rare
[36]. American basswood occurs in moist canyons in Nebraska that appear to be
refuges from fire [35]. Also in Nebraska, American basswood and other hardwoods
are more common than they were 130 years ago, and it is thought that
this is related to a decrease in the frequency and severity of fires
[60]. In New England northern hardwood forests that include American basswood
typically have fire-free periods on the order of 800 to more than 2,000
years [25].
Many authors report that hardwood forests including American basswood are
encroaching onto former grasslands since fires have been suppressed
[1,3,38,60,72]. In Iowa oak savanna is replaced by dense sugar
maple-American basswood forest process in approximately 200 years without fire
[56]. In Wisconsin American basswood are invading mixed oak stands from which
they had formerly been excluded by wildfire [13]. However, Auclair and
Cottam [5] stated that this is not a general trend; succession to sugar
maple-American basswood is confined to specific sites largely because of
fragmentation of forests and the resultant lack of seed sources. They
do concur that red oak stands may represent sugar maple-American basswood sites
that have either been retrogressed to oak by fire, or from which sugar
maple and American basswood have been excluded by fire [5].
A stand of American basswood and eastern hophornbeam in Minnesota contained
scattered open-grown, large bur oaks. The American basswood and eastern
hophornbeam were even-aged. Sugar maple was not present in the dominant
layer even though its presence would normally be expected. The forest
structure was explained by Daubenmire [19] as a sugar maple-American basswood
stand that had experienced a brief period of burning. Daubenmire
proposed the following relationship of sugar maple-American basswood forests to
fire: 1) sugar maple is eliminated by single fires; 2) repeated fires
eliminate elms and red oak and leave American basswood and eastern hophornbeam as
sprout thickets; 3) continued fires can eliminate American basswood and eastern
hophornbeam thickets, which are replaced by bur oak and grasses; 4)
severe fires will eventually eliminate bur oak, leaving prairie. The
structure of the forest described above was explained as the result of
sugar maple-American basswood-eastern hophornbeam invasion of a bur oak savanna
(possibly stage 3). The sugar maple forest developed to maturity
without experiencing fire. This forest then experienced a short period
of burning (perhaps only one fire) and had reached stage 2 when burning
ceased, leaving the American basswood and eastern hophornbeam to grow up into an
even-aged canopy. Daubenmire concluded that the climatic limits of
sugar maple-American basswood are further west than the actual limits due to fire
[19]. In Iowa oak savanna is replaced by dense sugar maple-American basswood
forest, the process occurring over the course of approximately 200 years
without fire [56].
Mature sugar maple-American basswood forests are very resistant to burning.
Decomposition of potential fuels is rapid, particularly on base-rich
mull soils. Dense shade reduces the numbers and cover of shrubs and
herbaceous species, and therefore very little fuel exists at ground
level. The tree trunks are not very flammable, and the open crowns do
not carry fire well. Low solar radiation, high relative humidity, and
low wind speeds enhance the moisture retention of ground-level fuels,
thereby inhibiting ignition and/or spread of fire. Ordinarily, only the
leaf litter ever reaches a flammable state, creating conditions
conducive only to patchy, creeping surface fires [32].
The Minnesota Big Woods (sugar maple-American basswood cover type) is often
described as an old, stable, climax forest. New evidence suggests that
in fact, these woods are of comparatively recent origin [65]. Climate
factors (increased moisture and lower temperatures) contributed to a
natural decrease in the frequency and intensity of fire. Once
established, sugar maple-American basswood forests do not burn easily [32],
creating conditions in which these forests were able to survive climatic
warming and increases in fire frequency. The extent of these woods has
been greatly reduced by human activities, but they have also experienced
lower danger from fire since humans have stopped setting fires and have
been active in suppressing fires [65].
American basswood occurs in aspen forests which are of postfire origin and
fire-maintained. The fire regime in these woods usually consists of
short fire intervals (on the order of 10 years) with low intensity
surface fires. Where these forests are protected from fire, closed
canopies and higher species diversity develop [37].
FIRE REGIMES:
Find fire regime information for the plant communities in which this
species may occur by entering the species name in the FEIS home page under
"Find Fire Regimes".
POSTFIRE REGENERATION STRATEGY:
Tree with adventitious-bud root crown/soboliferous species root sucker
FIRE EFFECTS
SPECIES: Tilia americana
IMMEDIATE FIRE EFFECT ON PLANT:
In Illinois a prescribed fall fire was conducted to open up areas that
had previously been classified as oak savanna. The low intensity fire
burned into adjacent closed canopy sugar maple-American basswood forest. Fuel
loading in the closed forest was approximately 530 g/sq m. Mortality of
American basswood stems under 4 inches in diameter (10 cm) was close to 10
percent (some of these were apparently only top-killed). Larger stems
were apparently unaffected by the fire [4].
DISCUSSION AND QUALIFICATION OF FIRE EFFECT:
NO-ENTRY
PLANT RESPONSE TO FIRE:
A prescribed fire in an aspen stand in which American basswood occurred
top-killed most of the trees in the stand. Those stems not killed by
the fire were felled to eliminate overstory competition. All of the
top-killed American basswood stems sprouted (stems ranged in size from 4 to 17
inches [10-43 cm] in diameter), producing an average of 21 sprouts per
clump within 5 years of the fire [57].
Fire wounding of American basswood increases susceptibility to butt rot [15]: of
trees with basal fire wounds, 100 percent of American basswood stems had butt
rot, resulting in a cull rate of 39 percent [39].
Light surface fires favor sugar maple seedlings over American basswood (and other
hardwoods). Hotter fires destroy existing reproduction of sugar maple
and create openings favoring American basswood. American basswood sprouts are less
abundant in stands escaping fire for extremely long periods of time,
presumably because the heavy shade created by very dense stands is not
tolerated by American basswood reproduction [50].
DISCUSSION AND QUALIFICATION OF PLANT RESPONSE:
The Research Project Summary Effects of surface fires in a mixed red and
eastern white pine stand in Michigan and the Research paper by Bowles
and others 2007 provide information on prescribed fire and postfire response
of several plant species, including American basswood, that was not available when
this species review was written.
FIRE MANAGEMENT CONSIDERATIONS:
The high heat value of American basswood wood is reported as 8,342 Btu per pound,
the low heat value averages 7,817 Btu per pound. The rate of fire
spread under laboratory conditions for American basswood wood is higher than that
in white fir (Abies concolor), sugar maple, southern magnolia (Magnolia
grandiflora), and longleaf pine (Pinus palustris). American basswood contains an
oil that is rich in volatile fatty acids [26].
Small American basswood slash, up to 2 inches (5 cm) in diameter, is usually very
quick to rot; that of most northern hardwood species, including
American basswood, is almost completely decayed in 4 to 7 years. Under average
conditions, American basswood logs and stumps are not a serious source of fire
danger after 10 to 12 years, and after 15 to 18 years, nothing remains
except mounds of moldy wood [62]. American basswood logs and slash are easily
water saturated, however, which slows the rate of decay [64].
Fuel values for herbicide-killed American basswood (as firewood) have been
reported [12].
Prescribed fire is not recommended for established stands of hardwoods
in which American basswood occurs; basal fire wounds increase susceptibility to
butt rot [39]. Use of fire in cut stands may improve resistance to butt
rot in the next generation of American basswood, since fire lowers the level at
which stump sprouts form [57].
REFERENCES
SPECIES: Tilia americana
REFERENCES:
1. Ahlgren, C. E. 1974. Effects of fires on temperate forests: north
central United States. In: Kozlowski, T. T.; Ahlgren, C. E., eds. Fire
and ecosystems. New York: Academic Press: 195-223. [13110]
2. Aikman, John M. 1926. Distribution and structure of the forests of
eastern Nebraska. University Studies. 26(1-2): 1-75. [6575]
3. Antrobius, William; Tagestad, Arden; Brumbaugh, Craig; Kresl, Steve.
1992. Integrated pest management & forest health for Sullys Hill
National Game Preserve. Report No. 91-11. Missoula, MT: U.S. Department
of Agriculture, Forest Service, Region 1 Timber, Cooperative Forestry
and Pest Management. 21 p. [18429]
4. Apfelbaum, Steven I.; Haney, Alan W. 1990. Management of degraded oak
savanna remnants in the upper Midwest: preliminary results from three
years of study. In: Hughes, H. Glenn; Bonnicksen, Thomas M., eds.
Restoration `89: the new management challenge: Proceedings, 1st annual
meeting of the Society for Ecological Restoration; 1989 January 16-20;
Oakland, CA. Madison, WI: The University of Wisconsin Arboretum, Society
for Ecological Restoration: 280-291. [14705]
5. Auclair, Allan N.; Cottam, Grant. 1971. Dynamics of black cherry (Prunus
serotina Erhr.) in southern Wisconsin oak forests. Ecological
Monographs. 41(2): 153-177. [8102]
6. Barrett, John W.; Ketchledge, Edwin H.; Satterlund, Donald R., eds.
1961. Forestry in the Adirondacks. Syracuse, NY: Syracuse University,
State University College of Forestry. 139 p. [21405]
7. Beck, Donald E. 1988. Regenerating cove hardwood stands. In: Smith, H.
Clay; Perkey, Arlyn W.; Kidd, William E., Jr., eds. Guidelines for
regenerating Appalachian hardwood stands: Workshop proceedings; 1988 May
24-26; Morgantown, WV. SAF Publ. 88-03. Morgantown, WV: West Virginia
University Books: 156-166. [13943]
8. Bovey, Rodney W. 1977. Response of selected woody plants in the United
States to herbicides. Agric. Handb. 493. Washington, DC: U.S. Department
of Agriculture, Agricultural Research Service. 101 p. [8899]
9. Braun, E. Lucy. 1936. Forests of the Illinoian till plain of
southwestern Ohio. Ecological Monographs. 6(1): 91-149. [8379]
10. Buell, Murray F.; Cantlon, John E. 1951. A study of two forest stands in
Minnesota with an interpretation of the prairie-forest margin. Ecology.
32(2): 294-316. [3251]
11. Carey, Andrew B. 1983. Cavities in trees in hardwood forests. In: Davis,
Jerry W.; Goodwin, Gregory A.; Ockenfeis, Richard A., technical
coordinators. Snag habitat management: proceedings of the symposium;
1983 June 7-9; Flagstaff, AZ. Gen. Tech. Rep. RM-99. Fort Collins, CO:
U.S. Department of Agriculture, Forest Service, Rocky Mountain Forest
and Range Experiment Station: 167-184. [17833]
12. Cassens, Daniel L.; Fischer, Burnell C. 1980. Fuel values of chemically
deadened white oak and basswood trees. Forest Products Journal. 30(8):
38-39. [22211]
13. Clark, Bryan F. 1970. Measures necessary for natural regeneration of
oaks, yellow-poplar, sweetgum, and black walnut. In: The silviculture of
oaks and associated species: A summary of current information:
Proceedings, Society of American Foresters annual meeting; 1968 October;
Philadelphia, PA. Res. Pap. NE-144. Upper Darby, PA: U.S. Department of
Agriculture, Forest Service, Northeastern Forest Experiment Station:
1-16. [13261]
14. Clark, James S. 1990. Landscape interactions among nitrogen
mineralization, species composition, and long-term fire frequency.
Biogeochemistry. 11(1): 1-22. [17185]
15. Collingwood, G. H. 1937. Knowing your trees. Washington, DC: The
American Forestry Association. 213 p. [6316]
16. Crow, T. R. 1990. Tilia americana L. American basswood. In: Burns,
Russell M.; Honkala, Barbara H., technical coordinators. Silvics of
North America. Volume 2. Hardwoods. Agric. Handb. 654. Washington, DC:
U.S. Department of Agriculture, Forest Service: 784-791. [21826]
17. Curtis, John T. 1959. The vegetation of Wisconsin. Madison, WI: The
University of Wisconsin Press. 657 p. [7116]
18. Dansereau, Pierre. 1959. The principal plant associations of the Saint
Lawrence Valley. No. 75. Montreal, Canada: Contrib. Inst. Bot. Univ.
Montreal. 147 p. [8925]
19. Daubenmire, Rexford F. 1936. The "big woods" of Minnesota: its
structure, and relation to climate, fire, and soils. Ecological
Monographs. 6(2): 233-268. [2697]
20. Davidson, Walter H. 1988. Potential for planting hardwoods in the
Appalachians. In: Smith, H. Clay; Perkey, Arlyn W.; Kidd, William E.,
Jr., eds. Guidelines for regenerating Appalachian hardwood stands:
Workshop proceedings; 1988 May 24-26; Morgantown, WV. SAF Publ. 88-03.
Morgantown, WV: West Virginia University Books: 255-268. [13951]
21. Dugger, Katie M.; Fredrickson, Leigh H. 1992. Life history and habitat
needs of the wood duck. Fish and Wildlife Leaflet 13.1.6. Waterfowl
Management Handbook. Washington, DC: U.S. Department of the Interior,
Fish and Wildlife Service. 8 p. [20789]
22. Duncan, Wilbur H.; Duncan, Marion B. 1988. Trees of the southeastern
United States. Athens, GA: The University of Georgia Press. 322 p.
[12764]
23. Erdmann, Gayne G. 1987. Methods of commercial thinning in even-aged
northern hardwood stands. In: Nyland, Ralph D., editor. Managing
northern hardwoods: Proceedings of a silvicultural symposium; 1986 June
23-25; Syracuse, NY. Faculty of Forestry Miscellaneous Publication No.
13 (ESF 87-002); Society of American Foresters Publication No. 87-03.
Syracuse, NY: State University of New York, College of Environmental
Science and Forestry: 191-210. [10657]
24. Eyre, F. H., ed. 1980. Forest cover types of the United States and
Canada. Washington, DC: Society of American Foresters. 148 p. [905]
25. Fahey, Timothy J.; Reiners, William A. 1981. Fire in the forests of
Maine and New Hampshire. Bulletin of the Torrey Botanical Club. 108:
362-373. [9707]
26. Fons, W. L.; Clements, H. B.; Elliott, E. R.; George, P. M. 1962.
Project Fire Model. Summary Progress Report-II. Period May 1, 1960, to
April 30, 1962. Macon, GA: U.S. Department of Agriculture, Forest
Service, Southeastern Forest Experiment Station, Southern Forest Fire
Laboratory. 58 p. [16824]
27. Frelich, Lee E.; Calcote, Randy R.; Davis, Margaret B.; Pastor, John.
1993. Patch formation and maintenance in an old-growth hemlock-hardwood
forest. Ecology. 74(2): 513-527. [21171]
28. Garrison, George A.; Bjugstad, Ardell J.; Duncan, Don A.; [and others].
1977. Vegetation and environmental features of forest and range
ecosystems. Agric. Handb. 475. Washington, DC: U.S. Department of
Agriculture, Forest Service. 68 p. [998]
29. Godfrey, Robert K. 1988. Trees, shrubs, and woody vines of northern
Florida and adjacent Georgia and Alabama. Athens, GA: The University of
Georgia Press. 734 p. [10239]
30. Godman, Richard M.; Mattson, Gilbert A. 1976. Seed crops and
regeneration problems of 19 species in northeastern Wisconsin. Res. Pap.
NC-123. St. Paul, MN: U.S. Department of Agriculture, Forest Service,
North Central Forest Experiment Station. 5 p. [3715]
31. Greller, Andrew M. 1988. Deciduous forest. In: Barbour, Michael G.;
Billings, William Dwight, eds. North American terrestrial vegetation.
Cambridge; New York: Cambridge University Press: 288-316. [19544]
32. Grimm, Eric C. 1984. Fire and other factors controlling the Big Woods
vegetation of Minnesota in the mid-nineteenth century. Ecological
Monographs. 54(3): 291-311. [22170]
33. Hansen, Henry L.; Kurmis, Vilis. 1972. Natural succession in
north-central Minnesota. In: Aspen: Symposium proceedings; [Date of
conference unknown]; Duluth, MN. Gen. Tech. Rep. NC-1. St. Paul, MN:
U.S. Department of Agriculture, Forest Service, North Central Forest
Experiment Station: 59-66. [12040]
34. Hardin, Kimberly I.; Evans, Keith E. 1977. Cavity nesting bird habitat
in the oak-hickory forests--a review. Gen. Tech. Rep. NC-30. St. Paul,
MN: U.S. Department of Agriculture, Forest Service, North Central Forest
Experiment Station. 23 p. [13859]
35. Harrison, A. Tyrone. 1980. The Niobrara Valley Preserve: its
biogeographic importance and description of its biotic communities. A
working report to the Nature Conservancy. 116 p. On file with: U.S.
Department of Agriculture, Forest Service, Intermountain Research
Station, Fire Sciences Laboratory, Missoula, MT. [5736]
36. Heinselman, Miron L. 1973. Fire in the virgin forests of the Boundary
Waters Canoe Area, Minnesota. Quaternary Research. 3: 329-382. [282]
37. Heinselman, Miron L. 1981. Fire intensity and frequency as factors in
the distribution and structure of northern ecosystems. In: Mooney, H.
A.; Bonnicksen, T. M.; Christensen, N. L.; [and others], technical
coordinators. Fire regimes and ecosystem properties: Proceedings of the
conference; 1978 December 11-15; Honolulu, HI. Gen. Tech. Rep. WO-26.
Washington, DC: U.S. Department of Agriculture, Forest Service: 7-57.
[4390]
38. Hendrickson, William H. [n.d.]. Perspective on fire and ecosystems in
the United States. In: [Publication unknown]: 29-33. On file with: U.S.
Department of Agriculture, Forest Service, Intermountain Research
Station, Fire Sciences Laboratory, Missoula, MT. [17276]
39. Hepting, George H.; Hedgcock, George G. 1935. Relation between butt rot
and fire in some eastern hardwoods. Tech. Note 14. Asheville, NC: U.S.
Department of Agriculture, Forest Service, Appalachian Forest Experiment
Station. 2 p. [10186]
40. Johnson, Paul S. 1976. Modal development of regeneration in clearcut red
oak stands in the Driftless Area. In: Fralish, James S.; Weaver, George
T.; Schlesinger, Richard C., eds. Central hardwood forest conference:
Proceedings of a meeting; 1976 October 17-19; Carbondale, IL.
Carbondale, IL: Southern Illinois University: 455-475. [3817]
41. Kartesz, John T.; Kartesz, Rosemarie. 1980. A synonymized checklist of
the vascular flora of the United States, Canada, and Greenland. Volume
II: The biota of North America. Chapel Hill, NC: The University of North
Carolina Press; in confederation with Anne H. Lindsey and C. Richie
Bell, North Carolina Botanical Garden. 500 p. [6954]
42. Kelty, Matthew J. 1988. Sources of hardwood regeneration and factors
that influence these sources. In: Smith, H. Clay; Perkey, Arlyn W.;
Kidd, William E., Jr., eds. Guidelines for regenerating Appalachian
hardwood stands: Workshop proceedings; 1988 May 24-26; Morgantown, WV.
SAF Publ. 88-03. Morgantown, WV: West Virginia University Books: 17-30.
[13931]
43. Kittredge, J., Jr. 1934. Evidence of the rate of forest succession on
Star Island, Minnesota. Ecology. 15(1): 24-35. [10102]
44. Kuchler, A. W. 1964. Manual to accompany the map of potential vegetation
of the conterminous United States. Special Publication No. 36. New York:
American Geographical Society. 77 p. [1384]
45. Lamson, Neil I. 1983. Precommercial thinning increases diameter growth
of Appalachian hardwood stump sprouts. Southern Journal of Applied
Forestry. 7(2): 93-97. [12563]
46. Lamson, Neil I. 1988. Role of stump sprouts in regenerating Appalachian
hardwood stands. In: Smith, H. Clay; Perkey, Arlyn W.; Kidd, William E.,
Jr., eds. Guidelines for regenerating Appalachian hardwood stands:
Workshop proceedings; 1988 May 24-26; Morgantown, WV. SAF Publ. 88-03.
Morgantown, WV: West Virginia University Books: 31-37. [13932]
47. Little, Elbert L., Jr. 1979. Checklist of United States trees (native
and naturalized). Agric. Handb. 541. Washington, DC: U.S. Department of
Agriculture, Forest Service. 375 p. [2952]
48. Loehle, Craig. 1988. Tree life history strategies: the role of defenses.
Canadian Journal of Forest Research. 18(2): 209-222. [4421]
49. Loucks, O. L. 1959. A forest classification for the Maritime Provinces.
Proceedings, Nova Scotian Institute on Science. 25: 86-167. [15408]
50. Maissurow, D. K. 1941. The role of fire in the perpetuation of virgin
forests of northern Wisconsin. Journal of Forestry. 39(2): 201-207.
[3445]
51. Martin, Christian J.; MacMillan, Paul C. 1982. Seven years of forest
succession in Happy Valley, Jefferson County, Indiana. Indiana Academy
of Science. 92: 197-206. [10369]
52. Matlack, Glenn R. 1987. Diaspore size, shape, and fall behavior in
wind-dispersed plant species. American Journal of Botany. 74(8):
1150-1160. [28]
53. Monk, Carl D.; Imm, Donald W.; Potter, Robert L.; Parker, Geoffrey G.
1989. A classification of the deciduous forest of eastern North America.
Vegetatio. 80: 167-181. [9297]
54. Morris, L. A.; Mollitor, A. V.; Johnson, K. J.; Leaf, A. L. 1979. Forest
management of floodplain sites in the northeastern United States. In:
Johnson, R. Roy; McCormick, J. Frank, technical coordinators. Strategies
for protection & mgmt of floodplain wetlands & other riparian
ecosystems: Proceedings of the symposium; 1978 December 11-13; Callaway
Gardens, GA. Gen. Tech. Rep. WO-12. Washington, DC: U.S. Department of
Agriculture, Forest Service: 236-242. [4364]
55. Nyland, Ralph D.; Marquis, David A. 1979. Appropriate silvicultural
systems for northern hardwoods. In: North America's forests: gateway to
opportunity: Proceedings of the 1978 joint convention of the Society of
American Foresters and the Canadian Institute of Forestry. Washington,
DC: Society of American Foresters: 334-338. [10023]
56. Patterson, Rich. 1992. Fire in the oaks. American Forests. 98(11):
32-34, 58-59. [19958]
57. Perala, Donald A. 1974. Growth and survival of northern hardwood sprouts
after burning. Res. Note NC-176. St. Paul, MN: U.S. Department of
Agriculture, Forest Service, North Central Forest Experiment Station. 4
p. [7349]
58. Pitel, J. A.; Wang, B. S. P. 1988. Improving germination of basswood
(Tilia americana L.) seeds with gibberellic acid. Seed Science and
Technology. 16: 273-280. [11404]
59. Raunkiaer, C. 1934. The life forms of plants and statistical plant
geography. Oxford: Clarendon Press. 632 p. [2843]
60. Rothenberger, Steven J. 1989. Extent of woody vegetation on the prairie
in eastern Nebraska, 1855-1857. In: Bragg, Thomas B.; Stubbendieck,
James, eds. Prairie pioneers: ecology, history and culture: Proceedings,
11th North American prairie conference; 1988 August 7-11; Lincoln, NE.
Lincoln, NE: University of Nebraska: 15-18. [14012]
61. Sander, Ivan L.; Merritt, Clair; Tryon, E. H. 1981. Oak-hickory. In:
Choices in silviculture for American forests. Washington, DC: Society of
American Foresters: 23-29. [6525]
62. Scholtz, H. F. 1930. How long does hardwood slash remain a fire menace?.
Journal of Forestry. 28: 568. [16386]
63. Smith, H. Clay; Miller, Gary W. 1991. Releasing 75- to 80-year-old
Appalachian hardwood sawtimber trees: 5-year d.b.h. response. In:
McCormick, Larry H.; Gottschalk, Kurt W., eds. Proceedings, 8th central
hardwood forest conference; 1991 March 4-6; University Park, PA. Gen.
Tech. Rep. NE-148. Radnor, PA: U.S. Department of Agriculture, Forest
Service, Northeastern Forest Experiment Station: 402-413. [15327]
64. Spaulding, Perley; Hansbrough, J. R. 1944. Decay of logging slash in the
Northeast. Tech. Bull. No. 876. Washington, DC: U.S. Department of
Agriculture. 22 p. [14337]
65. Sprugel, Douglas G. 1991. Disturbance, equilibrium, and environmental
variability: What is 'natural' vegetation in a changing environment?.
Biological Conservation. 58: 1-18. [18674]
66. Stickney, Peter F. 1989. Seral origin of species originating in northern
Rocky Mountain forests. Unpublished draft on file at: U.S. Department of
Agriculture, Forest Service, Intermountain Research Station, Fire
Sciences Laboratory, Missoula, MT; RWU 4403 files. 7 p. [20090]
67. Tilghman, Nancy G. 1987. Deer populations and their impact on
regenerating northern hardwoods. In: Nyland, Ralph D., editor. Managing
northern hardwoods: Proceedings of a silvicultural symposium; 1986 June
23-25; Syracuse, NY. Faculty of Forestry Miscellaneous Publication No.
13 (ESF 87-002); Society of American Foresters Publication No. 87-03.
Syracuse, NY: State University of New York, College of Environmental
Science and Forestry: 286-297. [10660]
68. Tubbs, Carl H. 1979. Northern hardwood ecology. In: North America's
forests: gateway to opportunity: Proceedings of the 1978 joint
convention of the Society of American Foresters and the Canadian
Institute of Forestry. Washington, DC: Society of American Foresters:
329-333. [10022]
69. Twight, Peter A.; Minckler, Leon S. 1972. Ecological forestry for the
Northern hardwood forest. Washington, DC: National Parks and
Conservation Association. 12 p. [3508]
70. USDA Natural Resources Conservation Service. 2018. PLANTS Database,
[Online]. U.S. Department of Agriculture, Natural Resources Conservation Service
(Producer). Available: https://plants.usda.gov/. [34262]
71. Voss, Edward G. 1985. Michigan flora. Part II. Dicots
(Saururaceae--Cornaceae). Bull. 59. Bloomfield Hills, MI: Cranbrook
Institute of Science; Ann Arbor, MI: University of Michigan Herbarium.
724 p. [11472]
72. Wanek, Wallace James. 1967. The gallery forest vegetation of the Red
River of the North. Fargo, ND: North Dakota State University. 190 p.
Dissertation. [5733]
73. Weaver, J. E. 1960. Flood plain vegetation of the central Missouri
Valley and contacts of woodland with prairie. Ecological Monographs.
30(1): 37-64. [275]
74. Yeager, A. F. 1935. Root systems of certain trees and shrubs grown on
prairie soils. Journal of Agricultural Research. 51(12): 1085-1092.
[3748]
75. Vogel, Willis G. 1981. A guide for revegetating coal minesoils in the
eastern United States. Gen. Tech. Rep. NE-68. Broomall, PA: U.S.
Department of Agriculture, Forest Service, Northeastern Forest
Experiment Station. 190 p. [15575]
FEIS Home Page



